a Biography of Cancer – Siddhartha Mukherjee, published in 2010.

I was gifted this book last year and dove into it three months ago. I am not an expert in medical practices, and blessed to have avoided being a patient of this family of diseases. But I have friends and family who have suffered and some who have passed; I thought it could help me better understand the science and current treatment practices that I see. The author is an acclaimed medical doctor / researcher – his writing style is technical yet clear and straightforward, complemented by historical events and real-life examples.
In 2010 (published date of this book), an estimated six hundred thousand Americans, and more than seven million humans around the world died of cancer. In the United States, one in three women and one in two men will develop cancer during their lifetime. A quarter of all American deaths, and about 15 percent of all deaths worldwide, will be attributed to cancer. In some nations, cancer will surpass heart disease to become the most common cause of death.
What I learned: The recorded history of this family of diseases goes all the way back to the earliest writings and descriptions from 2000 years ago. For hundreds of years the medical profession tried to explain the occurrence of cancer because of “bad blood”, though they had no way to cure it. Early treatments involved crude surgeries that provided short term relief in a few cases. Nineteenth century discoveries about the structure of cells provided some early insights. It took later discoveries about the role of certain viruses, and the structure of chromosomes / genes that began to reveal the mechanisms that drove cancer cells to divide. To a large degree cancer arises from harmful genetic mutations, whether driven by viral disruptions or the introduction of carcinogens into the system.
At some point, carcinogens were linked to cancer cases; the earliest of these were discovered through studies of illness in young chimney sweeps in the 1800’s. Later, studies of links between smoking and incidences of cancer were initiated after smoking became popular in the early 1920s, driven initially by the return of WW1 Soldiers. It took over 60 years to establish well-documented correlations and convince government agencies to take action against industry sponsored advertising:
“We believe that the products we make are not injurious to health. We always have and always will cooperate closely with those whose task it is to safeguard public health. – “A Frank statement to Cigarette Smokers,” a full-page advertisement produced by the tobacco industry in 1954. And then, 35 years later:
“Certainly, living in America in the last half of the 20th century, one would have to be deaf, dumb and blind not to be aware of the associated dangers, real or imagined, of cigarette smoking. Yet the personal choice to smoke is…the same kind of choice as the driver who downed the beers, and then the telephone pole.” – Open letter from the tobacco industry, 1988.
Cancer screening methods became available in the 20th century, including Pap Smears (discovered by George Papanicolaou, a Greek cytologist at Cornell University) and mammograms among many others.
University research and clinical field trials: I had no appreciation for the scope of research and associated clinical trials that were conducted through universities and research hospitals, funded through federal grants as well as large fundraising efforts. Remember the Jimmy Fund which was started so many years ago? Support Cancer Care and Research at Dana-Farber and the Jimmy Fund – Donate to Dana-Farber and help fight cancer | Dana-Farber Cancer Institute
Clinical trials were generally initiated to prove new treatment options. They were also used to track the efficacy of existing treatment efforts. A large clinical trial in the 1960s that evaluated the established practice of radical surgical mastectomy (breast, muscles and lymph nodes) to treat breast cancers was found to be no more effective than simple mastectomy, or localized surgery followed by an after treatment of chemotherapy and or radiation therapy. As a result, the radical mastectomy is rarely, if ever performed by surgeons today. Widespread surgical applications were supplemented, and in some cases replaced by additional treatment options. Research efforts resulted in the development of targeted molecular drug treatments (chemotherapy) and radiation – designed to target and kill cancer cells, coupled with stem cell transplants to restore the immune systems after treatment.
“Randomized screening trials are bothersome. It takes ages to reach an answer, and these need to be large-scale projects to be able to answer the questions. [But…} there is no second-best option.” H.J. de Koning. Annals of Oncology, 2003.
A description of cancer: A lung cell resides in the left lung of a forty-year-old fire-safety-equipment installer. One morning in 1968, a minute sliver of asbestos from his equipment wafts through the air and lodges in the vicinity of that cell. His body reacts to the sliver with inflammation. The cells around the sliver begin to divide furiously, like a miniscule wound trying to heal, and a small clump of cells derived from the original cell arises at the site. In one cell in that clump an accidental mutation occurs in a RAS gene…. The cell containing the mutant gene is driven to grow more swiftly than its neighbors and creates a clump within the original clump of cells. It is not yet a cancer cell, but a cell in which uncontrolled cell division has partly been unleashed – cancer’s primordial ancestor. A decade passes. A small collection of RAS-mutant cells continues to proliferate, unnoticed, in the far periphery of the lung. The man (also) smokes cigarettes, and a carcinogenic chemical in tar reaches the periphery of the lung and collides with the clump of RAS-mutated cells. A cell in this clump acquires a second mutation of its genes, activating a second oncogene.
Another decade passes. Yet another cell is caught in the path of an errant x-ray and acquires another mutation, this time inactivating a tumor suppressor gene. …. Now a fatal march is on. The cells, with multiple mutations begin to outgrow their brethren. As the cells grow, they acquire additional mutations resulting in further adaptations for growth and survival. A few cells acquire mobility, can migrate through the lung tissue and enter the bloodstream, and eventually acquiring the capacity to survive in bone tissue. It represents the first metastasis of a tumor that originated in the lung. The man is occasionally short of breath, he feels a tingle of pain in the periphery of his lung. Occasionally he senses something moving under his ribcage when he walks. Another year passes. The man visits a physical and a CT scan is performed, revealing a rind like mass wrapped around the bronchus in the lung. A biopsy reveals lung cancer. A surgeon examines the man and the CT scan of the chest and deems the cancer inoperable. Three weeks later the man returns to the clinic complaining of pain in his ribs and his hips. A bone scan reveals metastasis to the pelvis and the ribs.
Intravenous chemotherapy is initiated. The cells in the lung tumor respond. The man soldiers through a punishing regimen of multiple cell-killing drugs. But during the treatment one cell in the tumor acquires yet another mutation that makes it resistant to the drug used to treat the cancer. Seven months after his initial diagnosis, the tumor relapses all over the body – in the lungs, bones and liver. On the morning of October 17, 2004, deeply narcotized on opiates in a hospital bed in Boston and surrounded by his wife and children, the man dies of metastatic lung cancer, a sliver of asbestos still lodged in the periphery of his lung. He is seventy-six years old. The genes, carcinogens, and the sequence of mutations are all certainly hypothetical. But the body at its center is real. This man was the first person to die under the care of the author during his fellowship in cancer medicine at Massachusetts General Hospital.
“I am not opposed to optimism, but I am fearful of the kind that comes from self-delusion.” Marvin Davis, in the New England Journal of Medicine, talking about the “cure” for cancer.
Cancer rates: Progress has been steady but very slow, as cancer cells have many defenses against traditional treatments, including accelerating mutations that preserve future generations of these cells. Much of the decline in mortality rates over the past 50 years has been driven by smoking cessation programs and reduced exposures to toxic chemicals. Childhood cancer mortality rates have dropped by 70% since 1970, largely due to advances in leukemia treatment. I lost a classmate in grade school due to leukemia; perhaps she would have survived if diagnosed in today’s world. While many cancer mortality rates have decreased, some cancers, such as pancreatic and brain cancers, still show increasing mortality rates. In the U.S. in 2025, lung and bronchus cancer are projected to cause the most deaths (124,730), followed by colorectal cancer (52,900), and pancreatic cancer (51,980). Overall, cancer is the second leading cause of death. US cancer rates and trends: how have cancer rates and mortality changed over time? | USAFacts
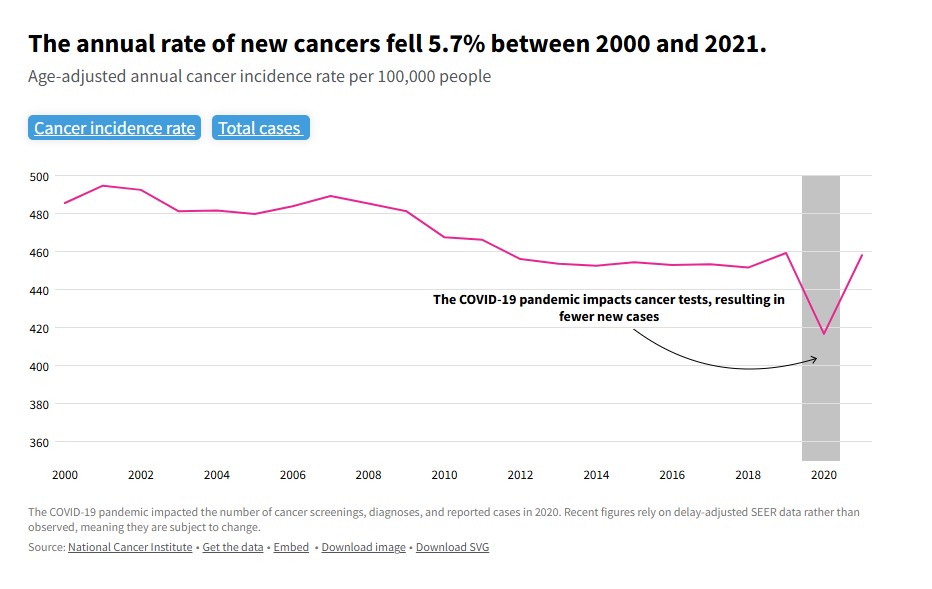
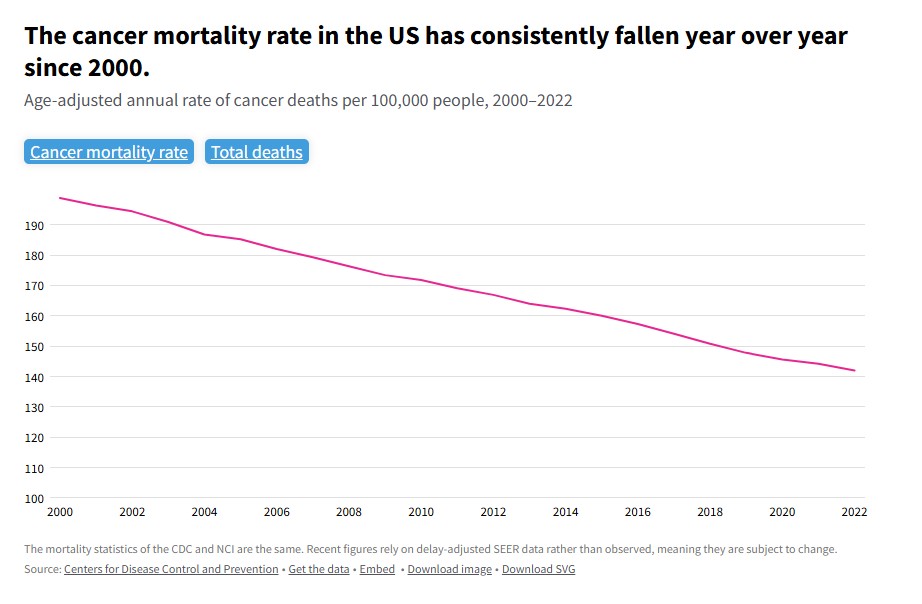
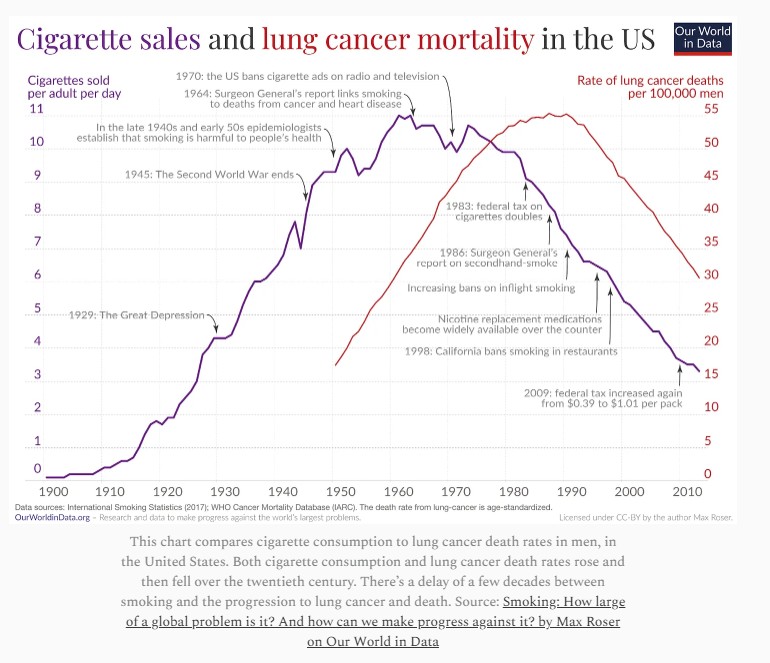
The decline in cancer mortality is about much more than smoking
“The National Cancer Institute, which has overseen American efforts on researching and combating cancers since 1971, should take on an ambitious and new goal…: the development of new drugs that will provide lifelong cures for many, if not all, major cancers. Beating cancer now is a realistic ambition because, at long last, we largely know it’s true genetic and chemical characteristics.” James Watson, 2009.
I have a greater appreciation for the science and research that has gone into fighting this disease, and the care of physicians for their patients, even now when I visit a cancer treatment center. Well done all.